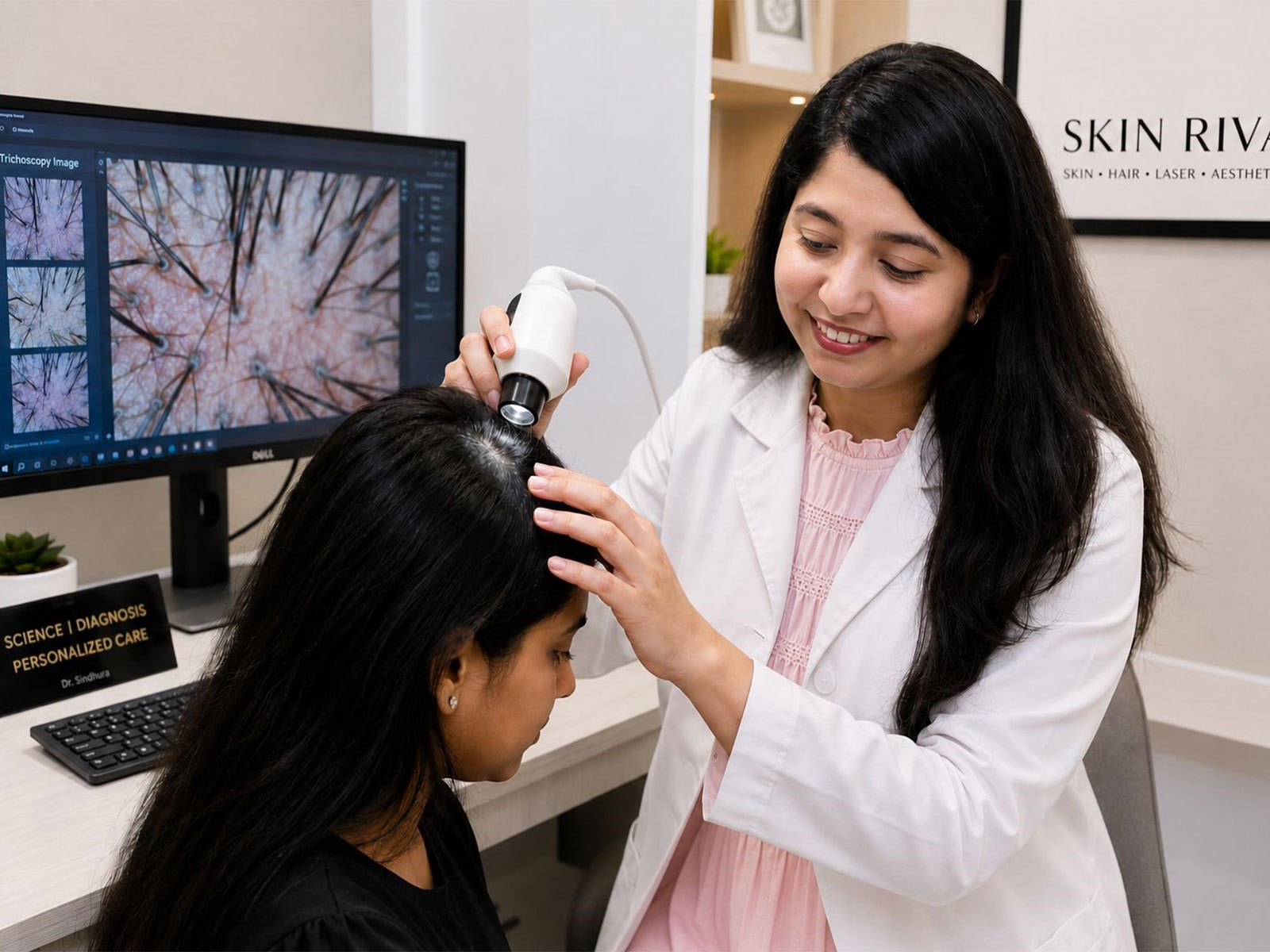
Abbey Road Institute Mumbai Strengthens Access to Music Production Education with Scholarship and Early Enrollment Support for August Intake
Jun 09, 2026
Mumbai (Maharashtra) [India], June 9: Abbey Road Institute Mumbai, the first Abbey Road Institute campus in Asia, has opened applications for its August 2026 intake of the 1-Year Advanced Diploma in Music Production & Sound Engineering, alongside a set of initiatives designed to support students before, during and after their training.
Read More...